
Lewis Structure - Nitrate anion
Тэги:
#Lewis_structure #nitrate_anion #chemistry #tutorialКомментарии:

This was really helpful~
Ответить
Very detailed explanation, thank you !!
Ответить
Finally I found a video that is just savage...thanks for this...I know I am watching it 12 years later but the explanation is just superb...😊😊
Ответить
I know it's 11 years later but may I just say - YOU ARE AN ANGEL SENT TO ME FROM OUR HEAVENLY FATHER. I have been searching for FOREVER for a video that thoroughly explains this in a way that I understand and now I understand!! Thank you thank you!!
Ответить
🌻🌻🌻
Ответить
thank you so much
Ответить
this is really helpful madam
Ответить
Thank u
Ответить
Thank u so much ... Finally this vdo helped me.. After watching a lot of vdo😊
Ответить
want to say it relay sucks
Ответить
👏✌️👏✌️👏✌️👌👍
Ответить
THANK YOU
Ответить
thanks
Ответить
Wow dis was the video which made me understood at last...phew!
Ответить
Thank you so much. I found a video about this for long time, this amazing 😊 helped me
Ответить
I saw many videos and this one was the only it really helped me. Thank u!!
Ответить
thnk u thnk u thnk uuuuu
Ответить
can i trust your teachings on this? lot of people randomly drawing it on the net.. who am i to trust
Ответить
Janet, is it correct to say the nitrate anion has sp² hybridization? Therefore the molecule is triangular planar?
Ответить
great explanation, there a few videos on this that are terrible, thanks
Ответить
That was very helpful!! Thanks for explaining the missing electron for nitrogen!! But how does it 'hops over' to Oxygen?
Ответить
thanks, that was very helpful!!!
Ответить
very cheerful video thanks LOL
Ответить
Thanks! Finally, this is not magic for me anymore.
Ответить
i swear YOU SAVED MY LIFE!!!
Ответить
So happy to find your videos so interesting. =D
Ответить
Actually, that's not true. She didn't show it in the video but this is actually known as dative bonding.... (there's supposed to be this weird arrow thingy she should have drawn, but she didn't....
Ответить
Brilliant!
Ответить
@BenJimi440 Yeah? Well how about you take some time off and make a video then? How else is the nitrate anion supposed to form, unless the nitrogen gives off an electron?
Ответить
I love solving equations the way you've taught - its fun. Can somebody give me more compounds to solve?
Ответить
I wish you were my chem teacher
Ответить
OMG thank you i was so lost but now i get it
Ответить
The last electron that you join should be a dative covalent bond
Ответить
These are really helpful. Thank you!
Ответить
WOW! Your lecture is really helpful for me! Thank You so much!!!
Ответить
Thanks this helped me learn how to draw lewis structures for polyatomic ions.
Ответить
thank you for your time and explanation. you are very helpful.
Ответить
thanks a lot, i really love your tutorial
Ответить
w0o0ow! thnx
Ответить
this is great, thank you so much.
Ответить
I have never benefitted from anything more than this in my life. Thanks a lot.
Ответить
very helpful...i have a question...so like what atoms can expand their octate??
Ответить
Thanks for making this video I had my head wrapped round it. Do you really need to indicate which atom has what charge? I mean don't U only need it when drawing Molecule shapes? with the partial positive and partial negative signs?
Ответить
you forgot to put a -1 after the brackets
Ответить
hahahaha i discobered one day before the exam ha-ha !!
Ответить
i have a final tomorrow and i dont even know what to do so lol this is really helping me thank u for doing this also cuz some pple dont explain it that good as u do and like this im getting it
Ответить
you saved my ass thank you for making this video
Ответить
youre smart =) cant wait to do this in chem
Ответить
why didnt i discover your video two days ago!!!!!!!!!!!!!! haha
Ответить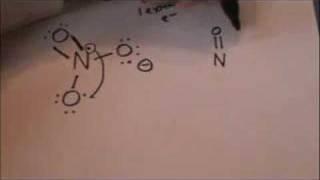
![[ASMR] Fortnite UNREAL RANK Gameplay Satisfying Keyboard 360 FPS [ASMR] Fortnite UNREAL RANK Gameplay Satisfying Keyboard 360 FPS](https://invideo.cc/img/upload/OGFJUjh2dUFhNDI.jpg)
























