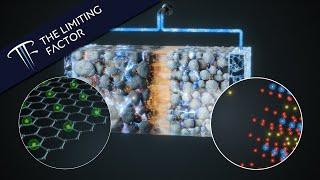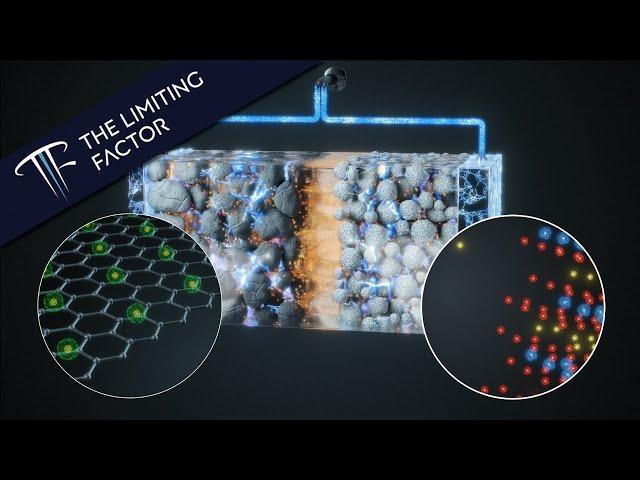
How a Lithium Ion Battery Actually Works // Photorealistic // 16 Month Project
Комментарии:

Interesting factoids and clarifications:
1) Although the electrons can travel at close to the speed of light, the rate at which the battery charges and discharges is often limited by the speed that lithium can travel between the electrodes through the electrolyte solution. When the battery is fully charged, the nickel oxide is starved of electrons, which can impact the lithium ion motion.
2) "Like a drop of ink dispersing in water." Lithium ions diffuse through a liquid, but there's a bit more going on here. The concentration gradient drives the lithium ions to the opposite electrode and is strengthened by the electrical field between the electrodes. That is, it's as if the ions are being pumped from one electrode to the other.
3) "When an electrical pathway opens between the anode and cathode, all the electrons between the cathode and anode sense the energy imbalance." What I'm referring to here is the complex dynamics of the electromagnetic fields that flow in and around the the electrical pathway between the cathode and anode. I anthropomorphised as a shortcut because electromagnetism would be another video entirely.
4) I used the chemistry definition of electropositive and electronegative in this video, which are different from the physics definition.

I'd like to know what is happening as the battery is losing it's juice, leading to it needing to be replaced. As in, how does the battery slowly die?
Ответить
This is an invaluable piece of content on the operation of the li ion batteries our world has used and will continue to use in to the future.
Thank you all for your hard work and contribution!

best video i've ever seen
Ответить
Did you create these models and graphics? If yes, what software did you use?
Ответить
Amazing! I was mesmerized from start to finish. Bravo!
Ответить
这是一个很好的工作,直观形象地让人们更好的理解锂离子电池。
Ответить
Omg... absolutely fantastic. This is great work.
Ответить
I don't have words to describe how good this video is. Thanks a lot.
Ответить
Please make a video like this about generators
Ответить
I have never seen any detailed explanation with such an amazing animated stuff. Superb work.
Ответить
wow.. I want to watch this on my HDTV while having a beer
Ответить
This video is incredible. Thank you so much, you have a new subscriber!!
Ответить
An excellent explanation and presentation! I think I'm beginning to understand. I wa a Navy ET, so have the basics down, but this is revelatory. Thank you.
Ответить
The best video I have ever seen. Fantastic!
Ответить
Please make a video on synthesis of metal organic frame work
Ответить
Great, I would like to use on my education website which is in the Kannada language, how to get permission?
Ответить
This was amazing. The sound queues, visuals and concepts altogether; truly a masterpiece!
Ответить
very nicely explained
Ответить
Fantastic video.
Ответить
Using graphite for the anode material has some drawbacks: it takes 6 carbon atoms to hold 1 lithium ion. But graphite is relatively stable and doesn't swell or shrink very much with repeated charge/discharge cycles. Recent work to increase energy density is using silicon anodes, which can hold 6 times more lithium, but the silicon swells up and shrinks substantially, leading to cracking of the anode. But it is to be hoped that this is just an engineering problem that can be solved without too much difficulty.
Ответить
Thanks!
Ответить
Mind blowing animation 👌
Ответить
Thx for this well presented video. I get an overview of the function in Li-ion batteries. That's all I need for now. So thank you again 😊
Ответить
Such videos are priceless for getting deep knowledge on an engineering/scientific scale. Thank you very much
Ответить
Wow! Which software have you used?
Ответить
you are a genius
Ответить
So boring omg 😢
Ответить
perfect
Ответить
well done thx
Ответить
This was such an amazing presentation. Thank you and I hope the video does well!
Ответить
This was a wonderful video, thank you for making and publishing it!
Ответить
Stored energy density will drive the eclectic car industry. Excellent video
Ответить
Thank you for this amazing content
Ответить
incredible
Ответить
Yesterday I made a short circuit by mistake with an 18650 battery taken out of a Lidl drill and it instantly caught fire in my hand, instinctively I threw it down but it had already caused a 5mm burn on my finger. These 18650 batteries are extremely dangerous, I never thought that from a short circuit a battery could catch fire in less than 1 second and become incandescent
Ответить
This was so so good . Love this so so much . Thanks so much
Ответить
Awesome
Ответить
I can't believe this is available , this video helps me to understand the complexity of the Li-ion Batteries
Ответить
That was awesome! Thank you! My best wishes from Brazil
Ответить
Really well explained! Thank you for this!
Ответить
wonderful
Ответить
😮
Ответить
THAT was cool. I teach high school science and will be showing this video every year. Awesome video, and multidisciplinary too, which matters when trying to teach kids "why" I'm teaching what I'm teaching. The infographic style is great. Hats off to your graphic arts team, and the science that drove the video was fascinating. Well explained! You have good teachers.
Ответить
If the response is "Positive"... LOL, see what he did there ;-)
Ответить
What a beautiful animation and information, Excellent job!
Ответить