Drawing Lewis Structures: Resonance Structures - Chemistry Tutorial
Комментарии:

Lots of thanks for your Videos
PS: Drawing Lewis Structures for Molecules- Considering Formal Charge doesn't work it says it's private ?

but there's no bracket?
Ответить
Very informative video but you should do something to reduce the sibilance created when you speak. You make a lot of hissing noise.
Ответить
I really enjoyed this tutorial .. thanx :)
Ответить
Thank you Dear :)
Ответить
YOUR VOICE IS SOOOOOO ANNOYING
Ответить
Thanks :)
Ответить
Thanks SOOO much I got it instantly when you explained it but when my lecturer explain it made no sense .... the paint eg works well :D
Ответить
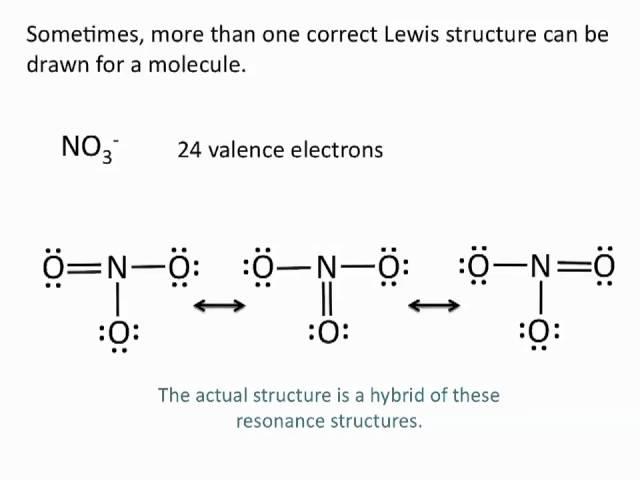
Your Lewis structure for sulfur dioxide is incorrect. There are two double bonds between both oxygen atoms and the sulfur. Remember that sulfur is in period three which means that it can break the octet rule and hold more than 8 electrons. There are no resonance structure for sulfur dioxide.
Ответить
Hello how do we know which atom is the central atom when starting to draw the lewis structure? for example what is the central atom is this caes CH3NHCH2CH3 ?
Ответить
how to draw a resonance structure for 1-methylcyclohexa-1,3-diene
Ответить
Thank you!
Ответить
Thanks! Helps a lot!!!
Ответить
your voice is the cutest thing to ever exist
Ответить
No, it means the absence of an electron
Ответить
voice is awesome..!!:p
Ответить
One thing that would be good to mention is the Electroneutrality Principle when doing any Lewis structure.
Ответить
Thank you a lot i had a lot of difficulty understanding this :)
Ответить
Very helpful, thank you!
Ответить
THANK YOU :D
Ответить